How representational incompetence and Johnstone’s triangle explain the silent collapse of understanding in advanced science classrooms.
Dr Caleb Moyo.
Introduction: The Real Problem Isn’t Ability, It’s Representation
Every A-level chemistry teacher has met them: bright, motivated students who can memorise equations and recite definitions and yet fall apart when asked to explain why a reaction occurs, how electrons move, or what a graph actually means. Their problem is not laziness or lack of intelligence; it is representational incompetence.
Representational incompetence refers to a learner’s inability to correctly interpret, translate, or coordinate the multiple forms in which scientific knowledge is expressed, such as symbols, diagrams, graphs, equations, and particle models (Kozma & Russell, 2005; Airey & Linder, 2009). Chemistry is uniquely vulnerable because it operates in three different representational worlds simultaneously, a reality first formalized by Johnstone (1991) in his famous Triangle of Chemical Representation.
Learning is disrupted when students are unable to move fluently between these worlds. Exams punish this mercilessly.
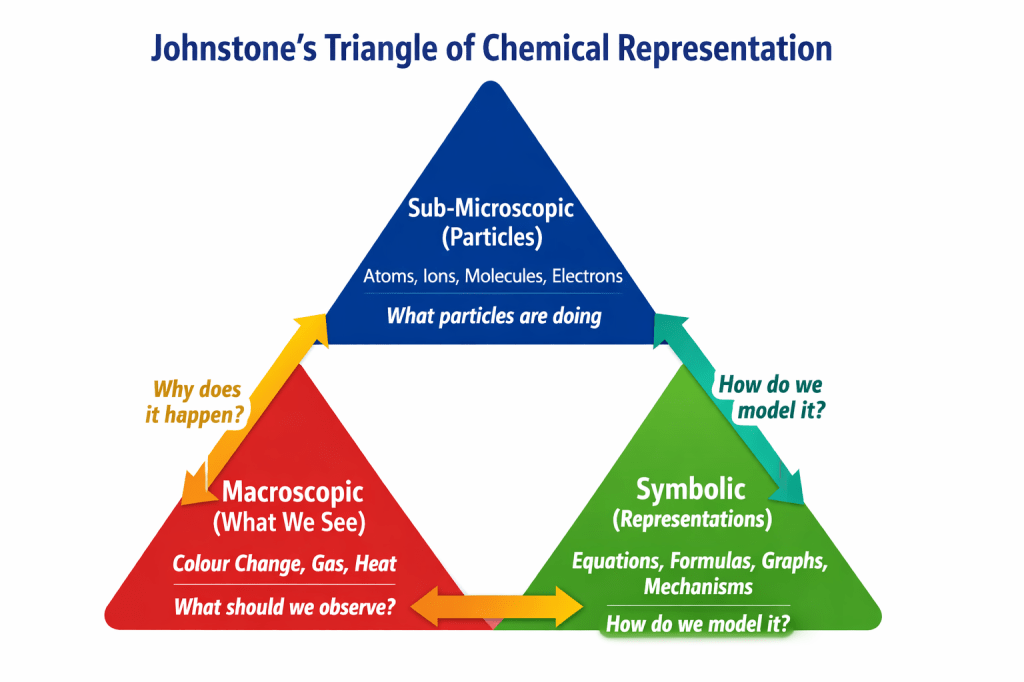
Johnstone’s Triangle: The Hidden Architecture of Chemistry Learning
All chemical understanding exists in three linked domains:
🟥 Macroscopic (What we observe)
Students can observe and measure color changes, precipitates, temperature increase, and gas bubbles.
🟦 Sub-microscopic (What particles do)
Atoms, ions, molecules, electrons, collisions, and intermolecular forces—nothing visible, everything explanatory.
🟩 Symbolic (How chemistry is written)
Equations, formulas, graphs, energy diagrams, rate laws, and mechanisms.
The fatal mistake in chemistry education.
Most A-level teaching occurs almost entirely in the symbolic corner, whereas students think in the macroscopic. Submicroscopic bridges are rarely built.
Johnstone (1991) showed that cognitive overload occurs when students must process all three types of information simultaneously without explicit guidance. Modern eye-tracking and interview studies have confirmed this (Ainsworth, 2006; Chandrasegaran et al., 2007).
Illustrative Diagram: Johnstone’s Triangle.
Research Insight.
Students do not naturally integrate macroscopic, sub-microscopic, and symbolic representations of concepts. Without explicit scaffolding, multiple representations increase cognitive load rather than reduce it (Johnstone, 1991; Ainsworth, 2006). When students fail, it is usually because one of these links is missing or broken.
Synthesis: Examiner Reports Confirm the Research.
Across all major A-level specifications, examiners report that students can recall equations and procedures but fail to interpret representations correctly, particularly ionic equations, energy diagrams, graphs, and organic mechanisms. This mirrors decades of chemistry education research on representational incompetence.Top of FormBottom of Form When examiner reports are read alongside the research literature, a striking alignment emerges:
| Examiner Observation | Research Explanation |
| Equations rewritten without meaning | Symbolic overload (Johnstone, 1991) |
| ΔH confused with Ea | Graph-as-picture error (Beichner, 1994) |
| Curly arrows misused | Mechanism memorisation (Bhattacharyya & Bodner, 2005) |
| Graph axes ignored | Representational decoding failure (McDermott et al., 1987) |
Examiners do not complain about careless students. They document the consequences of representational incompetence.
Why Representational Incompetence Is So Powerful.
Large-scale studies have shown that students can succeed algorithmically while failing conceptually.
Nakhleh and Mitchell (1993) found that students who scored well on stoichiometry tests could not explain reactions at the particle level. Bhattacharyya and Bodner (2005) found that organic chemistry students memorised mechanisms without understanding electron flow.
This explains the following familiar phenomenon:
“They can do the calculation, but they don’t know what it means.”
This is representational incompetence.
Ionic Equations: When Symbols Hide Chemistry.
Consider this reaction:
Na₂CO₃(aq) + 2HCl(aq) → 2NaCl(aq) + CO₂(g) + H₂O(l)
Most students “convert” this into:
Na₂CO₃ + 2HCl → 2NaCl + CO₂ + H₂O ❌
They have removed states but have not considered particles.
The correct ionic equation is as follows:
CO₃²⁻(aq) + 2H⁺(aq) → CO₂(g) + H₂O(l)
Here, the chemistry is based on ions, not compounds.
Taber (2009) showed that many A-level students think that Na₂CO₃ exists as intact molecules in solution. Their symbolic representation never reaches the submicroscopic domain.
Teaching Fix.
Make students draw:
- CO₃²⁻ reacting with H⁺
- Na⁺ and Cl⁻ floating unchanged
Only then are the equations allowed.
Energetics: When Energy Diagrams Become Artworks.
Students draw beautiful energy profile diagrams and still answer the following:
“Exothermic reactions are faster.”
This is wrong. The rate depends on the activation energy, not ΔH.
Students confuse vertical drop (enthalpy change) with vertical height (activation barriers).
Beichner (1994) demonstrated that students read graphs as images rather than encoded data.
Teaching Fix.
Every energy diagram must be accompanied by the following:
- numerical Ea
- ΔH values
- written interpretation
Force symbolic → conceptual → macroscopic links.
Kinetics: Graphs Without Meaning.
The concentration-time graph increased.
Student:
“The rate increases.”
Wrong axis. Wrong quantity.
McDermott et al. (1987) showed students rarely read axes unless forced.
Teaching Fix.
Requires full verbal translation:
“The concentration increases because…”
This forces the symbolic to macroscopic reasoning.
Organic Mechanisms: Arrows Without Electrons.
Students draw SN1 and SN2 arrows like decoration.
However, the arrows indicate the movement of electron pairs (Bhattacharyya & Bodner, 2005).
If a student cannot narrate:
“This lone pair attacks this carbon because…”
They do not understand.
Why This Happens Cognitively.
Human working memory can process approximately 4–7 elements simultaneously (Johnstone, 1991). Chemistry often requires
- formula
- state
- charge
- particle type
- energy
- diagram
- equation
That is overload. Students simplify by memorising symbols.
However, this strategy fails when reasoning is required.
How Teachers Can Fix It.
1. Representational Scaffolding
Always teach:
macroscopic → sub-microscopic → symbolic
Do not begin with equations.
2. Translation Tasks
Use:
- “Turn this equation into particles”
- “Turn this graph into words”
3. Delayed Interleaving.
Do not teach Equilibrium and kinetics should not be taught back-to-back. Same representations, different meanings = interference, but teach empirical formula and water of crystalisation as they are related.
4. Diagnostic Questions.
Q: What determines the reaction rate?
A. ΔH
B. Ea
C. Both
D. Neither
Correct: B
Q: Why does NaCl not appear in the ionic equations?
Answer: Because Na⁺ and Cl⁻ do not change chemically.
Concluding remarks.
Johnstone’s Triangle shows a hard truth:
Most A-level chemistry teaching lives in the symbolic world, while most students think in the macroscopic world. Representational incompetence is not a student’s failure; it is a design failure.
If we explicitly teach students how to move between the following:
- what they see,
- what particles do,
- and how chemistry is written, followed by conceptual understanding.
Exam scores also follow. Chemistry is a language. It is time that we taught it properly.
References.
Ainsworth, S. (2006). DeFT: A conceptual framework for considering learning with multiple representations. Learning and Instruction, 16(3), 183–198.
DOI: https://doi.org/10.1016/j.learninstruc.2006.03.001
🔗 https://www.sciencedirect.com/science/article/pii/S0959475206000181
Airey, J., & Linder, C. (2009). A disciplinary discourse perspective on university science learning. Studies in Higher Education, 34(1), 27–46.
DOI: https://doi.org/10.1080/03075070802528277
🔗 https://www.tandfonline.com/doi/full/10.1080/03075070802528277
Beichner, R. J. (1994). Testing student interpretation of kinematics graphs. American Journal of Physics, 62(8), 750–762.
DOI: https://doi.org/10.1119/1.17449
🔗 https://aapt.scitation.org/doi/10.1119/1.17449
Bhattacharyya, G., & Bodner, G. M. (2005). “It gets me to the product”: How students propose organic mechanisms. Journal of Chemical Education, 82(9), 1402–1407.
DOI: https://doi.org/10.1021/ed082p1402
🔗 https://pubs.acs.org/doi/10.1021/ed082p1402
Chandrasegaran, A. L., Treagust, D. F., & Mocerino, M. (2007). Development of a two-tier multiple-choice diagnostic instrument for evaluating secondary school students’ ability to describe and explain chemical reactions using multiple levels of representation. Chemistry Education Research and Practice, 8(3), 293–307.
DOI: https://doi.org/10.1039/B7RP90006F
🔗 https://pubs.rsc.org/en/content/articlehtml/2007/rp/b7rp90006f
Johnstone, A. H. (1991). Why is science difficult to learn? Things are seldom what they seem to be. Journal of Computer Assisted Learning, 7(2), 75–83.
DOI: https://doi.org/10.1111/j.1365-2729.1991.tb00230.x
🔗 https://onlinelibrary.wiley.com/doi/10.1111/j.1365-2729.1991.tb00230.x
Kozma, R., & Russell, J. (2005). Students becoming chemists: Developing representational competence. In J. K. Gilbert (Ed.), Visualization in Science Education (pp. 121–145). Springer.
DOI: https://doi.org/10.1007/1-4020-3613-2_8
🔗 https://link.springer.com/chapter/10.1007/1-4020-3613-2_8
McDermott, L. C., Rosenquist, M. L., & van Zee, E. H. (1987). Student difficulties in connecting graphs and physics concepts. American Journal of Physics, 55(6), 503–513.
DOI: https://doi.org/10.1119/1.15104
🔗 https://aapt.scitation.org/doi/10.1119/1.15104
Nakhleh, M. B., & Mitchell, R. C. (1993). Concept learning versus problem solving: There is a difference. Journal of Chemical Education, 70(3), 190–192.
DOI: https://doi.org/10.1021/ed070p190
🔗 https://pubs.acs.org/doi/10.1021/ed070p190
Taber, K. S. (2009). Learning at the symbolic level: Students’ understanding of chemical equations. Chemistry Education Research and Practice, 10(3), 171–179.
DOI: https://doi.org/10.1039/B901200A
🔗 https://pubs.rsc.org/en/content/articlehtml/2009/rp/b901200a
Leave a comment